Introduction to Molecular Geometry
Welcome to the fascinating world of molecular geometry! Have you ever wondered how the shape of a molecule influences its properties and behavior? The arrangement of atoms plays a crucial role in everything from chemical reactivity to biological functions. Today, we’re diving into one particular molecule that has piqued the interest of scientists and students alike: HNO2, or nitrous acid.
Understanding HNO2 molecular geometry can shed light on its characteristics and applications. So grab your lab coat as we explore what makes this molecule unique, starting with its structure and extending to real-world implications!
What is HNO2?
HNO2, or nitrous acid, is a weak acid commonly encountered in chemistry. It exists primarily in aqueous solutions and plays a significant role in various chemical reactions.
This compound contains nitrogen, oxygen, and hydrogen. Its unique structure allows it to behave both as an acid and as a source of nitrite ions (NO-).
One notable feature of TECHNO is its instability; it tends to decompose into nitric oxide (NO) and other byproducts when left alone for extended periods. Despite this instability, it has important applications in organic synthesis and analytical chemistry.
In nature, nitrous acid can be formed through the reaction of nitrogen oxides with water. This process is essential for understanding atmospheric chemistry and pollution dynamics.
The Lewis Structure of HNO2
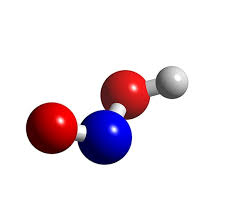
The Lewis structure of HNO2, or nitrous acid, is essential for understanding its molecular geometry. This diagram represents the arrangement of valence electrons among atoms.
In HNO2, nitrogen (N) is the central atom bonded to two oxygen (O) atoms and one hydrogen (H) atom. Each bond involves shared pairs of electrons that hold the molecule together.
One oxygen forms a double bond with nitrogen, while the other has a single bond along with an additional hydrogen ion attached. This setup highlights how electron distribution affects shape and polarity in molecules.
Dots are used to signify lone pairs on oxygen atoms. These unshared electrons play a crucial role in determining molecular behavior during chemical reactions.
Understanding this structure lays the groundwork for exploring HNO2’s properties and behavior in various applications.
Determining the Molecular Geometry of HNO2
To determine the molecular geometry of HNO2, we start by examining its Lewis structure. HNO2 consists of one hydrogen atom, one nitrogen atom, and two oxygen atoms.
The central nitrogen atom is bonded to both oxygen atoms. One of these bonds is a double bond with an oxygen while the other forms a single bond with another oxygen that carries a negative charge. This arrangement creates resonance structures which contribute to understanding the molecule’s shape better.
Using VSEPR (Valence Shell Electron Pair Repulsion) theory, we can predict how these bonds will arrange themselves in three-dimensional space. The presence of lone pairs on the nitrogen affects this geometry significantly.
HNO2 adopts a bent or angular shape due to repulsion between electron pairs around the nitrogen atom. This distinct configuration plays an essential role in its chemical properties and reactivity.
Factors Affecting Molecular Geometry
Molecular geometry is influenced by various factors that dictate how atoms arrange themselves in space. One major aspect is the presence of lone pairs on a central atom. These electrons exert repulsion, altering bond angles and overall shape.
Electronegativity also plays a crucial role. Atoms with higher electronegativity can pull shared electrons closer, affecting molecular polarity and spatial arrangement. This can lead to distortion in ideal geometric shapes.
Steric hindrance cannot be ignored either. Bulky groups around central atoms often create physical barriers that influence how closely other atoms can approach each other, leading to unexpected geometries.
Hybridization affects the orbital overlap between bonding atoms. Different types of hybrid orbitals will yield distinct molecular shapes based on their configurations and energy levels, further refining our understanding of structures like HNO2.
Real-Life Applications of Molecular Geometry
Molecular geometry plays a crucial role in various fields, from pharmaceuticals to materials science. Understanding the shape of molecules helps chemists design drugs that interact effectively with specific biological targets.
In agriculture, molecular geometry aids in developing pesticides that can attach more efficiently to pests while minimizing harm to beneficial organisms. This precision improves crop yields and promotes sustainable practices.
Furthermore, nanotechnology relies heavily on molecular geometry principles. Engineers manipulate molecular shapes to create new materials with desired properties, such as strength or conductivity.
Environmental science also benefits from this knowledge. By studying how pollutants interact at the molecular level, scientists devise strategies for effective remediation and pollution control.
In education, visualizing molecular structures enhances learning experiences. Students grasp complex concepts better when they can see how different geometries affect chemical behavior and reactions.
Conclusion
Understanding HNO2 molecular geometry is crucial for grasping its chemical behavior and reactivity. The unique arrangement of atoms in this molecule, which consists of nitrogen, oxygen, and hydrogen, plays a significant role in defining its properties. By analyzing the Lewis structure and applying VSEPR theory, one can determine that HNO2 has a bent shape due to the presence of lone pairs on the nitrogen atom.
Various factors influence molecular geometry, including electron repulsion and hybridization. These principles not only apply to HNO2 but extend across numerous molecules in chemistry. This understanding opens doors to real-life applications ranging from environmental science to pharmaceuticals.
As you delve deeper into molecular structures like HNO2, you’ll uncover their vast implications in various scientific fields. Whether you’re studying for an exam or simply curious about chemistry’s intricacies, knowing how molecular geometry influences compound behavior can enrich your knowledge significantly.